


By following the standard notation, you can avoid ambiguity in parsing and obtain accurate results. 110.984 g/mol The molar mass and molecular weight of CaCl 2 is 110.984. For example, C2H5OH for ethanol, H2O for water, or Na2SO4 for sodium sulfate.
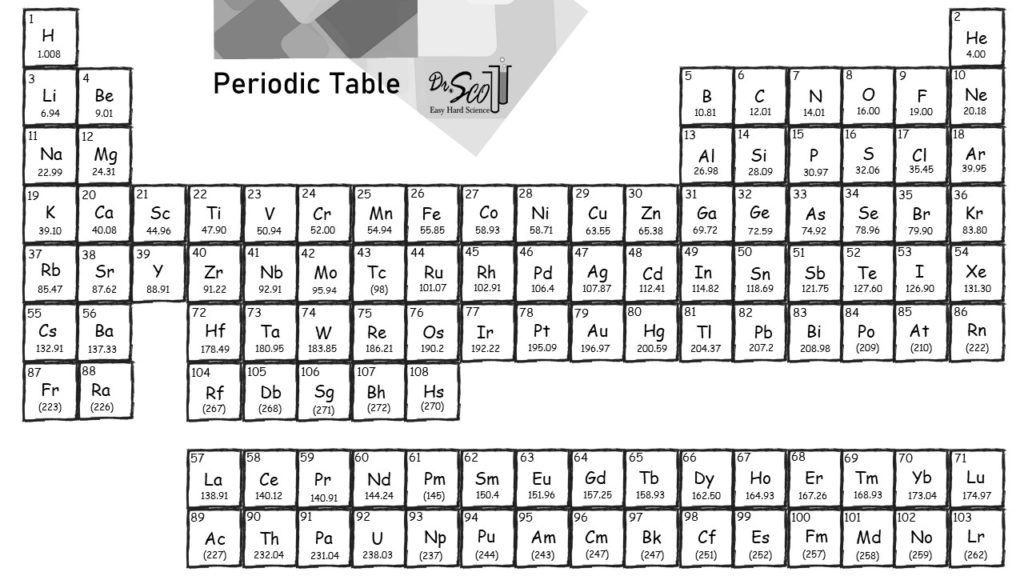
It is important to ensure that the chemical formula is entered correctly using standard notation. share my calculation Everyone who receives the link will be able to view this calculation. The result table includes the element symbol, atomic mass, multiplier (indicating the number of atoms of each element in the formula), and the calculated result. In addition to the molar mass, the calculator also provides a result table with detailed information on the individual elements in the formula. For example, when a non-electrolytic substance is dissolved in. The extent to which a substance associates or dissociates in a solution is described by the Van’t Hoff factor. It then calculates the molar mass by adding up the atomic weights of all the elements in the substance. the ratio of the concentration of particles formed when a substance is dissolved to the concentration of the substance by mass. The calculated value makes sense because it is almost four times times the mass for 1 mole of aluminum. The calculator utilizes the periodic table of elements to determine the atomic weights of the elements present in the formula. Prepare a concept map and use the proper conversion factor. To use the calculator, simply input the chemical formula of the substance and click the "Calculate" button. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in CaCl2: Molar Mass (g/mol) Ca (Calcium) 1 × 40.078 40.078. Then, lookup atomic weights for each element in periodic table: Ca: 40.078, Cl: 35. Molar mass is the mass of one mole of a substance and is expressed in grams per mole. Computing molar mass step by step First, compute the number of each atom in CaCl 2: Ca: 1, Cl: 2. Hence, molecular formula of CaCl 2 = 111 g/mol.The Molar Mass Calculator is a tool used to calculate the molar mass of a substance based on its chemical formula. There are 1 calcium atom and 2 chlorine atoms in this calcium chloride structure.The molecular formula of calcium chloride Calcium chloride consumption might result in hypercalcemia. Use this periodic table for calculating molar mass for any chemical formula. Consuming concentrated liquids or solid goods may result in gastrointestinal discomfort or ulcers. Formula: CaCl 2 Hill system formula: Ca 1 Cl 2 CAS registry number: 1 Formula weight: 110. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance. When solid calcium chloride dissolves exothermically, it can cause burns in the mouth and esophagus. Calcium chloride can irritate skin by desiccating it. It is a white powder or white-colored crystalline solid.Īlthough non-toxic in tiny doses when wet, the non-hydrated salt’s highly hygroscopic qualities pose considerable risks. To find the molar mass of a compound you would use the periodic table to find the molar mass of each element separately. This inorganic chemical is odorless and readily soluble in water. This article provides the calcium chloride formula, commonly known as the calcium dichloride formula or the calcium chloride anhydrous formula. The anhydrous salt is a desiccant since it is hygroscopic and deliquescent. These chemicals are mainly used for deicing and dust management. Molar mass of CaCl is 75.5310 g/mol Get control of 2022 Track your food intake, exercise, sleep and meditation for free. Calcium hydroxide can be used to neutralize hydrochloric acid.Ĭalcium chloride is a hydrated solid with the general formula CaCl 2xH 2 O, where x = 0, 1, 2, 4, and 6. At ambient temperature, it is a white crystalline solid that is readily soluble in water. Next, you have to find the molar mass of Ca (40.08 g/mol) and Cl (35.45 g/mol). In our case, our two elements are calcium and chlorine. Calcium chloride is an inorganic substance with the chemical formula CaCl 2, and it is a salt. 110.98g/ (mol) Let's start with the chemical formula of calcium chloride: CaCl2 To find the molar mass of a compound you would use the periodic table to find the molar mass of each element separately.
